Stronger Site Selection Starts With Better Investigator Data
SunshineMD empowers sponsors and CROs to make better site selection decisions before and during a trial by showing how U.S. clinical trial investigators have performed in real studies.
Investigator selection is one of the biggest sources of trial risk
Clinical teams select sites using feasibility responses, prior relationships and incomplete performance records.
SunshineMD provides comprehensive investigator performance history across studies, sponsors and therapeutic areas.
Better data leads to better site selection decisions—and fewer surprises once a trial is underway.
Questions about investigator data?

What is SunshineMD?
SunshineMD is an investigator intelligence database built for U.S. pharmaceutical clinical trials.
Using publicly reported data from the Physician Payments Sunshine Act (Open Payments) and ClinicalTrials.gov, SunshineMD cleans, reconciles and enriches investigator and study data to create a near-census view of U.S. clinical trial investigators dating back to 2013.
The Sunshine Score
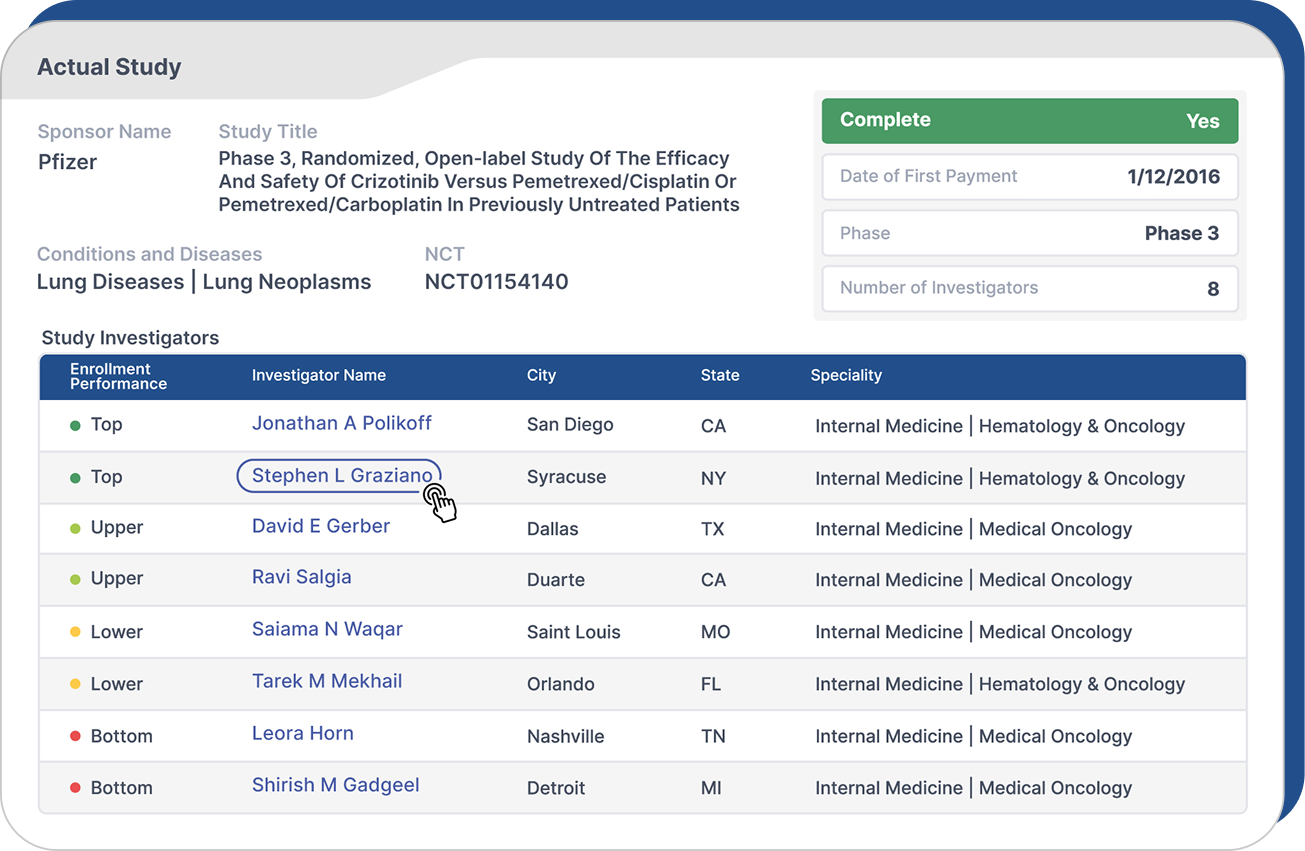
The Sunshine Score benchmarks investigators based on historical enrollment activity.
It gives teams a consistent way to compare performance, prioritize sites and flag risk during feasibility and site selection.
Insights teams gain with SunshineMD

Which investigators have experience in a specific indication or therapeutic area

How investigators have enrolled patients across prior studies

Which sites are most likely to contribute meaningfully
Who uses SunshineMD
Clinical operations leaders
Fast-growth biotech organizations
Feasibility and site selection teams
Data Science and Analytics teams
Built on public data.
Designed for better decisions.
SunshineMD works exclusively with publicly reported, non-patient data, allowing teams to evaluate investigator performance without introducing additional privacy or compliance risk.
By cleaning, reconciling and structuring complex datasets, the database helps teams make decisions they can stand behind.
See SunshineMD
In Action
If you’re planning a U.S.-based clinical trial and want better visibility into investigator performance, SunshineMD can show you how it works.
Request a Demo to explore investigator experience and enrollment history relevant to your next study.